Two years after his wife, the mother of his twin sons, died of colorectal cancer at age 36, Billy Boyle is leading a Cambridge, U.K.-based diagnostics developer in launching a 1,400-patient clinical trial to assess the accuracy of its diagnostic technology in detecting the disease early, from samples of breath and urine.
Boyle is co-founder and CEO of Owlstone Medical, which has commenced the 1,400-patient InTERCEPT trial. The study will examine the company’s microchip Field-Asymmetric Ion Mobility Spectrometer (FAIMS) platform technology, a noninvasive chemical detection system designed to assess a patient’s metabolome by measuring volatile organic compound (VOC) biomarkers from breath, urine, and other bodily fluids.
VOCs are the end product of metabolic processes within a person’s cells and tissues, within their microbiome, and from their response to environmental exposures. Since VOC production is linked directly to metabolic activity, the compounds can be sampled quickly and noninvasively from breath, urine, or other bodily fluids. Changes in VOC concentrations take place at the very earliest stages of disease, so detection of the compounds can allow disease diagnosis before other physical symptoms become apparent.
“Over time, it’s a bit like getting a window into a much larger volume of blood,” Boyle told Clinical OMICs. “Once every minute, your entire recirculated blood flow goes around your body. By sampling the chemicals coming from breath over that time period, you have really quite unbelievable test sensitivity in terms of trying to detect disease.
“Breath gives you that real unique advantage in terms of the test sensitivity, but also from the patient perspective. It’s the patient’s compliance and acceptability which is really important, and has historically been a major issue in colorectal cancer where other tests, be it a fecal-based test, or a colonoscopy, are very unpleasant,” Boyle added.
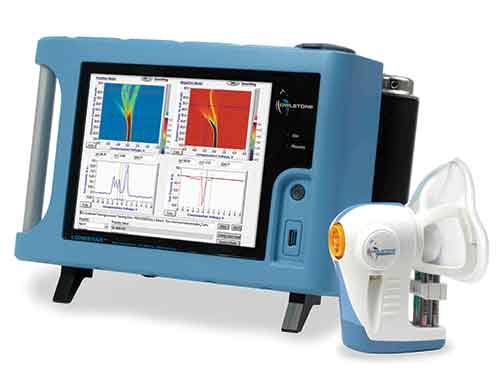
In the InTERCEPT trial, patients will have both their urine and breath tested, using Owlstone Medical’s ReCIVA breath sampler and Lonestar gas analyzer.
“We will work out which will give us the most optimal separation, and maximize the sensitivity and specificity. Or perhaps even a combination might improve it. We don’t know, and that has never been done before, which is why we wanted to test it in a large series,” said Ramesh P. Arasaradnam, Ph.D., principal investigator for InTERCEPT.
Dr. Arasaradnam is an associate professor of gastroenterology at the University of Warwick and a consultant gastroenterologist at University Hospital Coventry and Warwickshire NHS Trust. Both institutions are partnering with Owlstone in the trial, which is expected to take 12 to 18 months to complete.
InTERCEPT follows up on a 2014 pilot study assessing FAIMS in colorectal cancer detection. In the study, published in PLoS One, the technology showed a sensitivity of 88% in detecting VOC biomarkers for colorectal cancer in urine, and 62% sensitivity in detecting advanced adenomas in urine. Detection of VOCs in breath via FAIMS has also been pilot studied in conditions that include hepatic encephalopathy, inflammatory bowel disease and tuberculosis.
“The wide application in essence has given us confidence in the very technology, and certainly our methods. Also importantly, the bioinformatics, the actual analysis done in our collaboration with the engineers that we work with, all seem to work very well,” Dr. Arasaradnam said.
FAIMS is a variant of ion mobility spectrometry, a method of distinguishing charged gaseous molecules based on differences in the speed that they move through a buffer gas under the influence of an oscillating electric field.
Following ionization, VOC ions pass through channels across which an asymmetric radio frequency (RF) field is applied. Under the first portion of the waveform, ions will drift in one direction at a velocity based on their individual mobility in that electric field. As the field polarity is reversed, the ions change direction and speed. The mobility of the ions during the two parts of the waveform is rarely equal, resulting typically in a net drift towards one of the electrodes.
In FAIMS, this net drift is corrected for by applying an additional DC voltage or “compensation field,” focusing specific ions through the device to the detector. The FAIMS device produces a compensation field spectrum for the transmission of all ions.
Could the VOCs to be detected through InTERCEPT differ from one form of cancer to another?
“This is a great question. The short answer is, no one knows for sure,” Boyle said. “I think the kind of working hypothesis that exists today is that you have some general cancer metabolic markers associated with the growth of the tumor, but actually you will also almost certainly have site-specific volatile markers. In colorectal cancer, for instance, as the tumor grows in the colon, it starts to disrupt the local microbiome as well. And you see that reflected in changes in the metabolism as well.
“Most likely, what we’ll find is a combination of a general cancer marker with specific cancer type markers as well,” Boyle added.
To see more articles from the March/April issue of Cliniclal OMICs click here.